Acanthomorpha
G. David Johnson and E. 0. Wiley

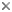
This tree diagram shows the relationships between several groups of organisms.
The root of the current tree connects the organisms featured in this tree to their containing group and the rest of the Tree of Life. The basal branching point in the tree represents the ancestor of the other groups in the tree. This ancestor diversified over time into several descendent subgroups, which are represented as internal nodes and terminal taxa to the right.

You can click on the root to travel down the Tree of Life all the way to the root of all Life, and you can click on the names of descendent subgroups to travel up the Tree of Life all the way to individual species.
For more information on ToL tree formatting, please see Interpreting the Tree or Classification. To learn more about phylogenetic trees, please visit our Phylogenetic Biology pages.
close boxIntroduction
Acanthomorpha, the spiny-rayed fishes, are the crown group of the Teleostei. Encompassing approximately 16,000 species in over 300 families (including the more familiar cods, tunas, flounders, basses, and perches as well as the exotic oarfishes, stonefishes, seahorses, flatfishes, puffers and much, much more), they comprise over 60% of extant teleosts and about one-third of all living vertebrates. Acanthomorphs exhibit staggering diversity in adult and larval body form, skeletal and soft anatomy, size, habitat, physiology, and behavior. They are found in most aquatic habitats available to fishes, from the abyssal plains of the ocean basins to mountain streams, desert springs, and caves and range in size from some of the longest (17 meters, the lampridiform, Regalecus glesne) and most massive (>3000 kilograms, the tetraodontiform, Mola mola) bony fishes to the smallest of all vertebrates (7-8mm, the gobioid, Schindleria) (Stiassny, et al., 2004; Nelson, 2006).
Characteristics
Synapomorphies
- True dorsal and anal fin spines present (Johnson and Patterson, 1993); there are notable exceptions, e.g., gadiforms, most lampridiforms, various perciforms).
- Median chondrified rostral cartilage present and strongly bound to premaxillary ascending process via well-developed rostro-premaxillary ligament (Hartel and Stiassny, 1986; Stiassny, 1986; Johnson and Patterson, 1993).
- Median caudal cartilages absent (Fujita, 1990; Johnson and Patterson, 1993).
- Anterior and medial infracarinales separate (Stiassny, 1993; Johnson and Patterson, 1993).
- Dorsal limb of posttemporal firmly bound to exoccipital (Stiassny, 1986; Johnson and Patterson, 1993)
- Medial pelvic processes ossified distally (Johnson and Patterson, 1993).
- First centrum with anterior surface bearing distinct facets that articulate with the exoccipital condyles (Rosen, 1985, Johnson and Patterson, 1993).
Discussion of Phylogenetic Relationships
The Acanthomorpha originated with Rosen's (1973) seminal paper on interrelationships of higher euteleosts. It comprised two putatively monophyletic clades, Paracanthopterygii and Acanthopterygii and was based on five ambiguously distributed characters. The monophyly of Rosen's Acanthomorpha, with Myctophiformes as their sister group, has subsequently been corroborated with morphological characters by Stiassny (1986), Hartel and Stiassny (1986), and Johnson and Patterson (1993), whereas monophyly of the two subgroups has been refuted (see discussion on Acanthopterygii page).
Molecular studies have challenged acanthomorph monophyly only in tending to place myctophiforms as the sister group of one or more of the most basal acanthomorph clades (e.g., Chen et al. 2003, Miya et al., 2003, 2005), for which there is no morphological support. The most extensive review and defense of acanthomorph monophyly was that of Johnson and Patterson (1993), emanating from their attempt to define the largest and most diverse acanthomorph assemblage, Percomorpha, in which they reviewed and evaluated support for previous hypotheses and proposed a morphology-based hypothesis of acanthomorph relationships. In so doing, they identified seven evolutionary innovations that unite the Acanthomorpha. Perhaps the most convincing of these are the presence in the dorsal and anal fins of true fin spines, and a single median chondrified rostral cartilage associated with specific rostral ligaments (Hartel and Stiassny, 1985; Stiassny, 1986) that permit the jaws to be greatly protruded while feeding. These two "key innovations" are also arguably those that have facilitated the greatest diversification in the evolution of this immense clade. Johnson and Patterson (1993) proposed a phylogeny for six basal acanthomorph groups leading sequentially to a newly defined Percomorpha.
References
Chen, W.-J., C., Bonillo, and G. Lecointre. 2003. Repeatability of clades as a criterion of reliability: a case study for molecular phylogeny of Acanthomorpha (Teleostei) with larger number of taxa. Mol. Phylo. Evol. 26(2):262-288.
Fujita, K. 1990, The caudal skeleton of teleostean fishes: Tokyo: Tokai University Press, xiii + 897 pp.
Hartel, K. E., and M. L. J. Stiassny. 1986. The identification of larval Parasudis (Teleostei: Chloropthalmidae); with notes on the anatomy and relationships of aulopiform fishes. Breviora 487:1-23.
Johnson, G. D. and C. Patterson. 1993. Percomorph phylogeny: A survey of acanthomorph characters and a new proposal. Bull. Mar. Sci. 52, 554-626.
Miya, M., H. Takeshima, H. Endo, N. B. Ishiguro, J. G. Inous, T. Mukai, T. P. Satoh, M. Yamagucki, A. Kawaguchi, K. Mabuchi, S. M. Shirai, and M. Nishida. 2003. Major patterns of higher teleost phylogenies: a new perspective based on 100 complete mitochondrial DNA sequences. Mol. Phylo. Evol. 26:121-138.
Miya, M., Satoh, T.P., and Nishida. 2005. The phylogenetic position of toadfishes (Order Batrachoidiformes)in the higher ray-finned fishes as inferred from partitioned Bayesian analysis of 102 whole mitochondrial sequences. Biol. Jour. Linn. Soc. 85:289-306
Nelson, J. S. 2006. Fishes of the World, 4th Edition. Wiley-Interscience, New York.
Rosen, D. E. 1973. Interrelationships of higher euteleostean fishes. In Interrelationships of Fishes. (Greenwood, P. H., Miles, S., Patterson, C. eds), pp 397-513. J. Linn. Soc. (London) 53 Supplement 1. Academic Press, New York, New York.
Rosen, D. E. 1985. An essay on euteleostean classification. Amer. Mus. Novit. 2827:1-57.
Stiassny, M. L. J. 1986. The limits and relationships of the acanthomorph fishes. J. Zool. London (B) 1:411-460.
Stiassny, M. L. J., Wiley, E. O., Johnson, G. D. & deCaravalho, M. R. 2004. Gnathostome fishes. In: Assembling The Tree of Life. ( J. Cracraft, ed.). Oxford University Press: 410-429.
Title Illustrations

| Scientific Name | Chilomycterus antennatus (Cuvier) (Diodontidae) |
|---|---|
| Location | Cayman Islands, Caribbean |
| Specimen Condition | Live Specimen |
| Identified By | Jeff Leis, Ichthyology, Australian Museum |
| Copyright |
© Greg and Marybeth Dimijian

|
| Scientific Name | Micropterus dolomieu |
|---|---|
| Comments | Smallmouth Bass |
| Creator | Eric Engbretson |
| Source Collection | U.S. Fish and Wildlife Service Online Digital Media Library |
About This Page

Smithsonian Institution, Washington, D. C., USA
E. 0. Wiley

University of Kansas, Lawrence, Kansas, USA
Correspondence regarding this page should be directed to G. David Johnson at and E. 0. Wiley at
Page copyright © 2006 and E. 0. Wiley
All Rights Reserved.
- First online 12 July 2005
- Content changed 09 January 2007
Citing this page:
Johnson, G. David and E. 0. Wiley. 2007. Acanthomorpha. Version 09 January 2007 (under construction). http://tolweb.org/Acanthomorpha/15088/2007.01.09 in The Tree of Life Web Project, http://tolweb.org/








 Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site
Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site